Executive Summary
Detailed intelligence on FDA CURRENT GOOD MANUFACTURING PRACTICES. Americanbible Data Intelligence synthesis of 10 verified sources complemented by 8 graphic references. This analysis also correlates with findings on Rare diseases: FDA proposes new approval system for therapies to provide a broader context. Unified with 9 parallel concepts to provide full context.
FDA CURRENT GOOD MANUFACTURING PRACTICES Overview and Information
Detailed research compilation on FDA CURRENT GOOD MANUFACTURING PRACTICES synthesized from verified 2026 sources.
Understanding FDA CURRENT GOOD MANUFACTURING PRACTICES
Expert insights into FDA CURRENT GOOD MANUFACTURING PRACTICES gathered through advanced data analysis in 2026.
FDA CURRENT GOOD MANUFACTURING PRACTICES Detailed Analysis
In-depth examination of FDA CURRENT GOOD MANUFACTURING PRACTICES utilizing cutting-edge research methodologies from 2026.
Everything About FDA CURRENT GOOD MANUFACTURING PRACTICES
Authoritative overview of FDA CURRENT GOOD MANUFACTURING PRACTICES compiled from 2026 academic and industry sources.
FDA CURRENT GOOD MANUFACTURING PRACTICES Expert Insights
Strategic analysis of FDA CURRENT GOOD MANUFACTURING PRACTICES drawing from comprehensive 2026 intelligence feeds.
Visual Analysis
Data Feed: 8 Units
IMG_PRTCL_500 :: FDA CURRENT GOOD MANUFACTURING PRACTICES

IMG_PRTCL_501 :: FDA CURRENT GOOD MANUFACTURING PRACTICES

IMG_PRTCL_502 :: FDA CURRENT GOOD MANUFACTURING PRACTICES

IMG_PRTCL_503 :: FDA CURRENT GOOD MANUFACTURING PRACTICES

IMG_PRTCL_504 :: FDA CURRENT GOOD MANUFACTURING PRACTICES
IMG_PRTCL_505 :: FDA CURRENT GOOD MANUFACTURING PRACTICES

IMG_PRTCL_506 :: FDA CURRENT GOOD MANUFACTURING PRACTICES
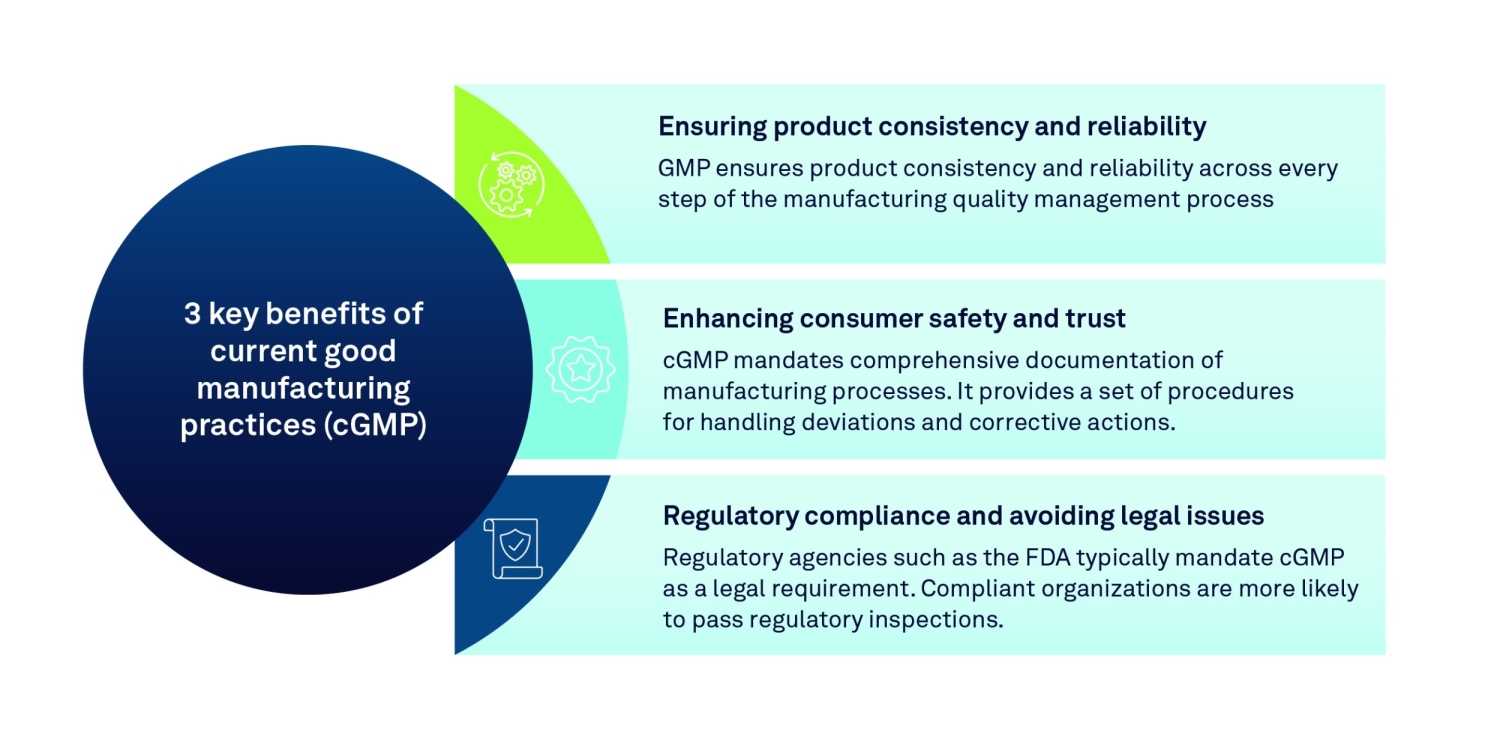
IMG_PRTCL_507 :: FDA CURRENT GOOD MANUFACTURING PRACTICES
Comprehensive Analysis & Insights
Research meticulous insights into fda current good manufacturing practices. This intelligence node has curated 10 intelligence streams and 8 distinct images. It is correlated to 9 related topics for deeper exploration.
Helpful Intelligence?
Our neural framework utilizes your validation to refine future datasets for FDA CURRENT GOOD MANUFACTURING PRACTICES.